Advancing Clinical Development with Clarity and Confidence
We partner with biotech, pharma, and healthcare innovators to translate science into measurable impact.
From early discovery to late-stage delivery, Alithia Life Sciences brings strategic clarity, operational precision, and commercial insight to every stage of your journey.

About Us
Alithia is a proudly Australian-owned, full-service CRO.
At Alithia, we understand that successful clinical trials require more than operational delivery, they require trusted partnership, strategic insight, and unwavering commitment.
Our team works collaboratively with clients to navigate the complexities of clinical development while maintaining the highest standards of quality, efficiency, and compliance.
Supported by experienced Senior Clinical research professionals, we bring deep expertise across multiple therapeutic areas, all phases of clinical research and experience in relevant regulatory jurisdictions.
With a proactive and solutions-focused approach, we are dedicated to delivering reliable outcomes and advancing programs with confidence from study start-up through to completion.
What We Do
We combine deep therapeutic expertise with executional excellence to help you move faster, smarter, and with greater confidence.
Turn complexity into clear direction.
- Provision of indicative estimates, timelines, and cost scenarios for planning purposes, investor and board discussions
- Regulatory and clinical development strategy
- Key Opinion Leader (KOL) engagement
- Clinical Trial Liaison services – your local support person
- Clinical development program optimisation
- Trial design and site strategy
- Project management and operational delivery including monitoring and site management services
- Vendor oversight and operational scale-up
- Enabling international sponsors to conduct clinical trials in Australia with confidence, whist meeting regulatory requirements
- Clear governance and experienced local oversight
We work across the life sciences ecosystem to help provide meaningful and globally acceptable data:
- Biotech – scaling from discovery through to all stages of clinical development execution
- MedTech and Digital Health – navigating complex regulatory landscapes and clinical trial delivery
- Pharma – optimising portfolios and accelerating program delivery
- Investors – providing cost and timeline scenarios, de-risking assets and unlocking value
- FIH and Healthy Volunteer studies
- Neurology
- Devices
- Endocrinology
- Oncology
- Women’s health
- Ophthalmology
- Gastroenterology
- Cardiovascular
- Respiratory
- Rare and Paediatric diseases
- Vaccines, and more
The Alithia team has extensive experience supporting a variety of Investigational Products in Drugs, Devices, Cell and Gene therapies and combination therapy trials.

Our Vision And Mission

Alithia partners with clients to deliver clinical development programs with speed, precision, and measurable impact, helping advance human health and wellbeing worldwide.
By combining agility, flexibility, and deep operational expertise, we adapt quickly to the evolving demands of clinical development and consistently deliver high-quality outcomes with efficiency and excellence.
Our commitment is simple:
- Deliver projects efficiently and successfully
- Add strategic value across every program
- Support better outcomes for human health and wellbeing
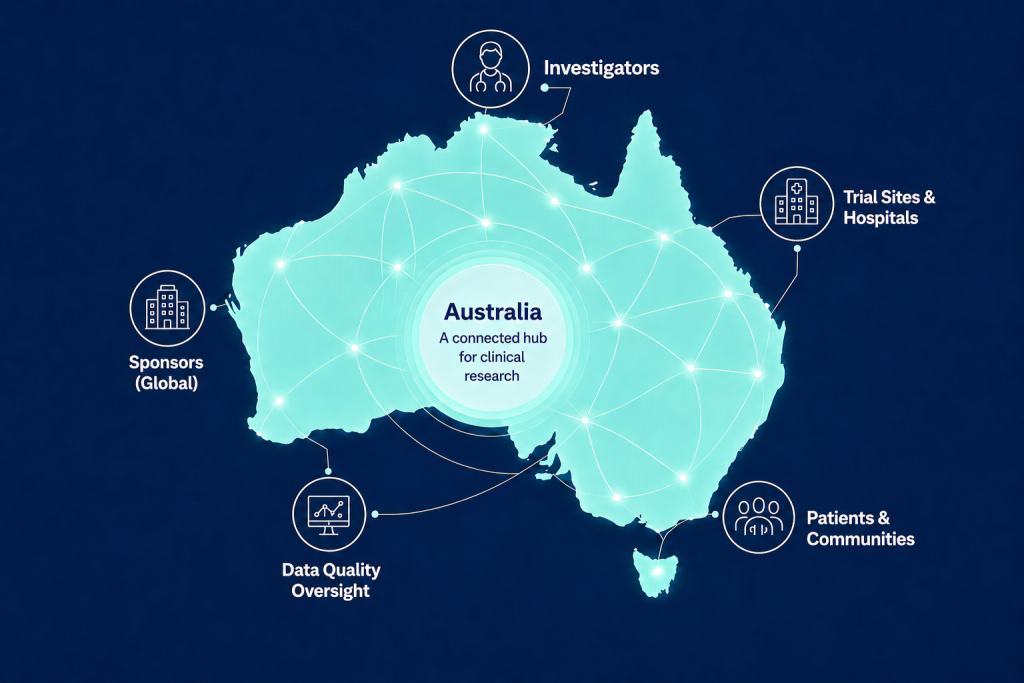
Why Australia?
Australia has long emerged as a premier destination for cutting-edge clinical research, offering a uniquely efficient and innovation-friendly environment for clinical trials.
- IND is not required for clinical trials
- GMP product not required for true FIH studies (for drug, devices across Phase I, II, III trials)
- Rapid start-up timelines through streamlined and well-developed Ethics process – from protocol to first patient (2-3 month on average)
- Up to 43.5% R&D cash tax incentive
- Globally recognised and accepted data (FDA, EMA)
- Centralised Site Contracts ensuring streamlined contracting process
- Diverse patients from an ethnically diverse background with over 30% of Australians born overseas and 50% of Australians with at least one parent born overseas
- KOLs and experienced site staff, underpinned by a world-class healthcare infrastructure
Alithia delivers FIH and Phase I studies with speed and precision, then seamlessly supports progression into Phase II and beyond.
From Australia and New Zealand to global multi-site programs through our trusted Global partner network, we ensure continuity across every stage of development. By retaining critical project knowledge and maintaining operational momentum, we help keep your timelines on track from start to finish.
Meet Our Leaders

A/PROF TINA SOULIS
Founder and Director
A/Prof Tina Soulis has over 30 years of working in the healthcare (pharmaceuticals and devices), biotechnology, academic research, clinical research and management sectors in senior roles including: CEO of a Contract Research Organisation, Director of a successful ASX200 company, VP of Clinical Strategy and Development with an innovative biotechnology company and now, Founder and Director of her own full-service capability clinical CRO.
A dynamic and strategic leader, Tina has an extensive network of stakeholders all over the world and experience in many therapeutic areas across all phases on development. Tina is one of the few professionals in Australia to have taken a product from concept to FDA approval.
MS SAMANTHA GEORGE
Director of Clinical Operations
A senior executive with over 28 years of experience in research and development, biotechnology and clinical trials sectors. Specialised in building trusted teams and stakeholder relationships, with expertise in developing structure, systems and processes for emerging and expanding organisations.
MS JOVANA MILOSEVIC
Chief Commercial Officer
Jovana is a distinguished clinical research executive with over 16 years of leadership experience spanning CRO management, clinical operations, business development, and key account management. She has held roles with leading global organisations, including IQVIA, Parexel, and Medtronic. She was most recently Head of a boutique Australian CRO. Her expertise extends across a broad range of therapeutic areas such as oncology, radiopharmaceuticals, GMOs, gastroenterology, cardiology, coronary and structural heart devices, infectious diseases, and dermatology.
Throughout her career, Jovana has successfully led clinical trials from FIH through post-marketing phases and has developed customised data-driven solutions to enhance post-marketing sales strategies. Her strategic approach, operational excellence, and deep cross-functional experience position her as a trusted leader in the clinical research and medical technology landscape.
Testimonials
Upcoming Events
Connect with us at major industry events across Australia and beyond to explore new ideas, partnerships, and possibilities.
BIOMELBOURNE NETWORK BIOFORUM
AI in Clinical Trials: Intelligence by Design
Gadens, Level 13 / 447 Collins Street
Melbourne, Australia
June 9, 2026, between 16:00 and 18:30
Learn more and register here
ARCS AUSTRALIA ANNUAL CONFERENCE 2026
International Convention Centre Sydney (ICC Sydney)
Sydney, Australia
June 10 – 12, 2026
Learn more and register here
EXPLORING NEW MODELS OF GLOBAL CLINICAL TRIAL COLLABORATION – CALIFORNIAN ROADSHOWS (USA)
Hosted by the Australian Trade and Investment Commission and MTPConnect, supported by Biocom
The Bay Area / San Francisco – June 17, 2026, 14:30-17:30 PDT
Learn more and register here
Los Angeles – June 18, 2026, 15:00-18:00 PDT
Learn more and register here
RESI SAN DIEGO 2026
JULEP Venue
San Diego, USA
June 22, 2026
Learn more and register here
BIO 2026
San Diego Convention Center
San Diego, USA
June 22 to 25, 2026
Learn more and register here


Contact Us
Let’s Start a Conversation
Whether you’re advancing a pipeline, or navigating complex challenges, we’re here to help.
Alithia Life Sciences: Overview | LinkedIn





















